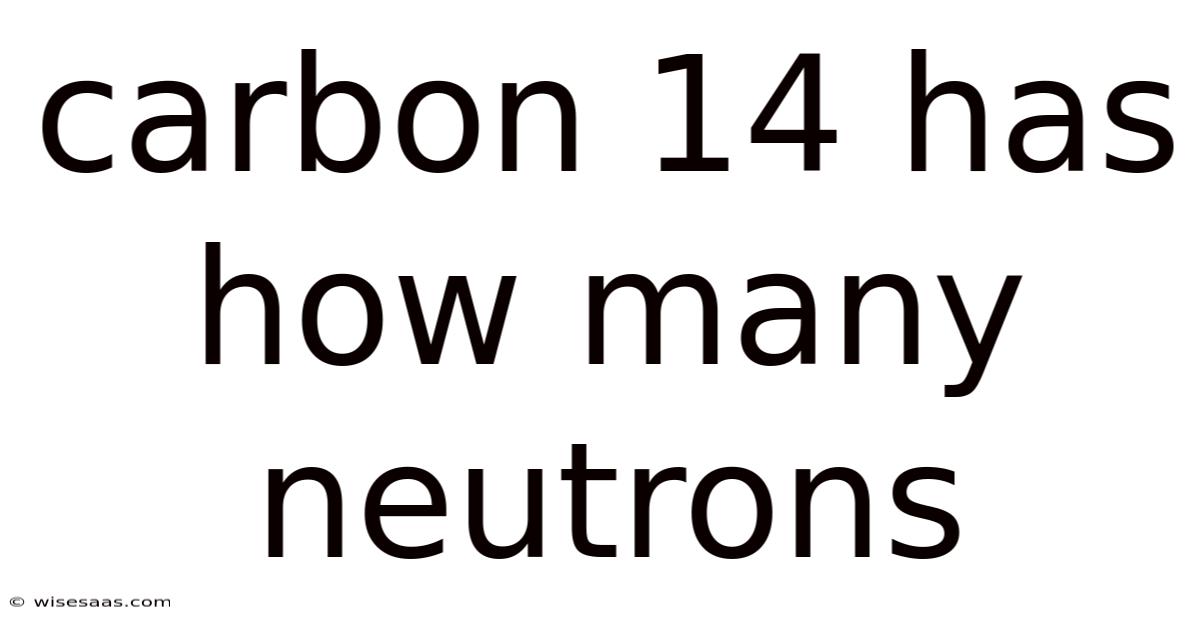Understanding the structure of carbon-14 and its properties is essential for grasping its role in radiocarbon dating and other scientific applications. Day to day, many people wonder about the specific details of this isotope, such as its neutron count, and how this impacts its behavior in nature. In this article, we will break down the fascinating world of carbon-14, exploring its atomic composition, its significance, and why its neutron count matters.
Carbon-14 is a radioactive isotope of carbon that matters a lot in various scientific fields, particularly in archaeology and geology. Because of that, this process creates a unique isotope that scientists use to date organic materials. But what exactly makes carbon-14 different from other carbon isotopes? It is formed in the upper atmosphere when cosmic rays interact with nitrogen atoms. The answer lies in its neutron count, which plays a vital role in its stability and behavior.
To understand carbon-14, we first need to look at its atomic structure. Here's the thing — carbon is an element with the atomic number 6, meaning it has six protons in its nucleus. Carbon-14 is a radioactive isotope, which means it undergoes radioactive decay over time. On the flip side, the number of neutrons—those not accounted for by protons—varies depending on the isotope. This decay process is what makes it valuable for dating purposes Worth keeping that in mind..
The neutron count in carbon-14 is 8. The half-life of carbon-14 is approximately 5,730 years, which means that after that time, half of the original amount of carbon-14 will have decayed into nitrogen-14. Think about it: this is significant because it influences how the isotope behaves in chemical reactions and its half-life. This characteristic is crucial for scientists to determine the age of samples that contain carbon-14.
Understanding the neutron count also helps us appreciate why carbon-14 is so important in modern science. The presence of eight neutrons in this isotope allows it to interact with other elements in unique ways, making it a reliable marker for time. Without this knowledge, the applications of carbon-14 in various fields would be limited Less friction, more output..
The official docs gloss over this. That's a mistake.
In addition to its role in dating, the neutron count affects how carbon-14 is produced and detected. Scientists rely on the presence of neutrons in this isotope to measure its concentration in the atmosphere and in samples. This understanding is essential for ensuring accurate results in radiocarbon dating, which is widely used to determine the age of artifacts and fossils And that's really what it comes down to..
This changes depending on context. Keep that in mind.
As we explore further, it becomes clear that the neutron count in carbon-14 is not just a number—it is a key factor that shapes its behavior and utility. This insight helps us appreciate the complexity of atomic structures and their impact on real-world applications. Whether you're a student, a researcher, or simply curious about science, understanding the role of neutrons in carbon-14 enhances your grasp of this important isotope.
The importance of carbon-14 extends beyond its neutron count. It serves as a bridge between the past and the present, allowing us to uncover secrets hidden within ancient materials. Practically speaking, by analyzing the neutrons in this isotope, scientists can reconstruct historical timelines and gain insights into the evolution of life on Earth. This connection between science and history underscores the value of studying atomic properties like neutron count It's one of those things that adds up..
At the end of the day, the neutron count in carbon-14 is a fundamental aspect of its identity. It influences how the isotope functions, how it is produced, and how it is utilized in various scientific endeavors. By understanding this detail, we gain a deeper appreciation for the detailed workings of the universe. But whether you're exploring the mysteries of time or the origins of life, the neutron count in carbon-14 remains a vital element in our quest for knowledge. This knowledge not only enhances our understanding of the past but also opens doors to future discoveries in science and technology And that's really what it comes down to. And it works..
The journey into the world of carbon-14 reveals much about the delicate balance of atomic forces. Day to day, each neutron plays a role in shaping the isotope's properties, making it a subject of fascination for researchers and students alike. Also, as we continue to explore this topic, we uncover the significance of these tiny particles in the grand tapestry of science. Understanding the neutron count in carbon-14 not only enriches our knowledge but also inspires curiosity about the mysteries of the natural world.
Beyond the sheer count of neutrons, the way those neutrons are arranged inside the carbon‑14 nucleus has practical consequences for how we detect the isotope. When a carbon‑14 atom decays, it emits a beta particle—a high‑energy electron—while the neutron count drops from eight to seven, converting the nucleus into nitrogen‑14. Consider this: the energy spectrum of those beta particles is directly tied to the nuclear binding energy, which itself depends on the neutron‑proton configuration. Modern decay‑counting instruments, such as liquid scintillation counters and proportional gas tubes, are calibrated to the specific beta‑energy profile of carbon‑14. If the neutron number were different, the decay energy would shift, forcing a redesign of detection hardware and altering the limits of sensitivity for very small samples.
This subtle interplay also shows up in atmospheric chemistry. By monitoring the neutron‑driven production rate, scientists can correct radiocarbon dates for these variations, a process known as calibration. Cosmic rays striking nitrogen‑14 atoms in the upper atmosphere knock out a neutron, turning the nitrogen into carbon‑14. This means the global inventory of carbon‑14 is not static; it waxes and wanes over decades. The rate at which this conversion happens is a function of the neutron flux in space, which fluctuates with solar activity and the Earth’s magnetic field. The internationally accepted calibration curves—such as IntCal20—are built upon a massive database of tree‑ring, coral, and lake‑sediment records that indirectly record past neutron fluxes. Without a precise grasp of how neutrons generate carbon‑14, these calibration tools would be far less reliable.
In the laboratory, the neutron count becomes a lever for creating carbon‑14 on demand. The efficiency of this reaction hinges on the neutron capture cross‑section of nitrogen‑14, which is itself a quantum‑mechanical property dictated by the arrangement of the eight neutrons. By optimizing beam energy and target composition, researchers can maximize carbon‑14 yield, supplying the isotope for medical diagnostics (e.g.Particle accelerators can bombard nitrogen‑14 targets with high‑energy particles, inducing a (n,p) reaction that swaps a neutron for a proton. , tracer studies in pharmacokinetics) and for environmental tracing (such as following carbon flow in ecosystems).
The practical upshot is that the neutron count is a linchpin connecting three realms: nuclear physics, geoscience, and applied technology. Plus, each neutron contributes to the stability of the carbon‑14 nucleus, governs the energy of its beta decay, and determines how readily the isotope can be produced or measured. This cascade of effects illustrates a broader principle in science: seemingly minute details at the subatomic level can ripple outward to shape entire fields of inquiry.
This is where a lot of people lose the thread.
Looking Ahead: Emerging Applications
As analytical techniques become ever more sensitive, the neutron signature of carbon‑14 may find novel uses. One promising avenue is “single‑molecule radiocarbon dating,” where researchers aim to date microscopic fragments of organic matter—such as individual pollen grains or micro‑fossils—by counting the discrete beta events they emit. In such experiments, the statistical noise is dominated by the stochastic nature of neutron‑mediated decay, making a deep understanding of neutron behavior essential for interpreting results It's one of those things that adds up..
Another frontier lies in climate reconstruction. Ice cores and deep‑sea sediments trap ancient carbon compounds that retain their original carbon‑14 content. By coupling high‑precision neutron‑count models with isotope‑ratio mass spectrometry, scientists can tease apart subtle variations in past atmospheric carbon cycles, offering new insights into how greenhouse gases have fluctuated over glacial‑interglacial timescales Easy to understand, harder to ignore..
Finally, in the realm of nuclear security, carbon‑14’s neutron profile is being explored as a forensic marker. Since anthropogenic nuclear events (e.So g. , detonations or reactor accidents) imprint distinctive neutron flux signatures on surrounding carbon reservoirs, measuring deviations in the neutron‑derived carbon‑14 ratios could help identify and date clandestine nuclear activities The details matter here..
Concluding Thoughts
The eight neutrons residing in carbon‑14 are far more than a static tally; they are active participants in a cascade of processes that enable us to peer into the deep past, monitor the present, and anticipate future scientific breakthroughs. Here's the thing — their influence stretches from the cosmic ballet that creates the isotope in the upper atmosphere, through the meticulous calibration of radiocarbon dates, to the cutting‑edge technologies that harness carbon‑14 for medicine, environmental science, and security. Recognizing the central role of neutron count transforms carbon‑14 from a simple dating tool into a multidisciplinary bridge linking atomic physics with the narratives of Earth’s history. As we continue to refine our measurement techniques and expand our theoretical models, the humble neutron will remain a key that unlocks new chapters in our understanding of both the natural world and the technologies we devise to explore it.