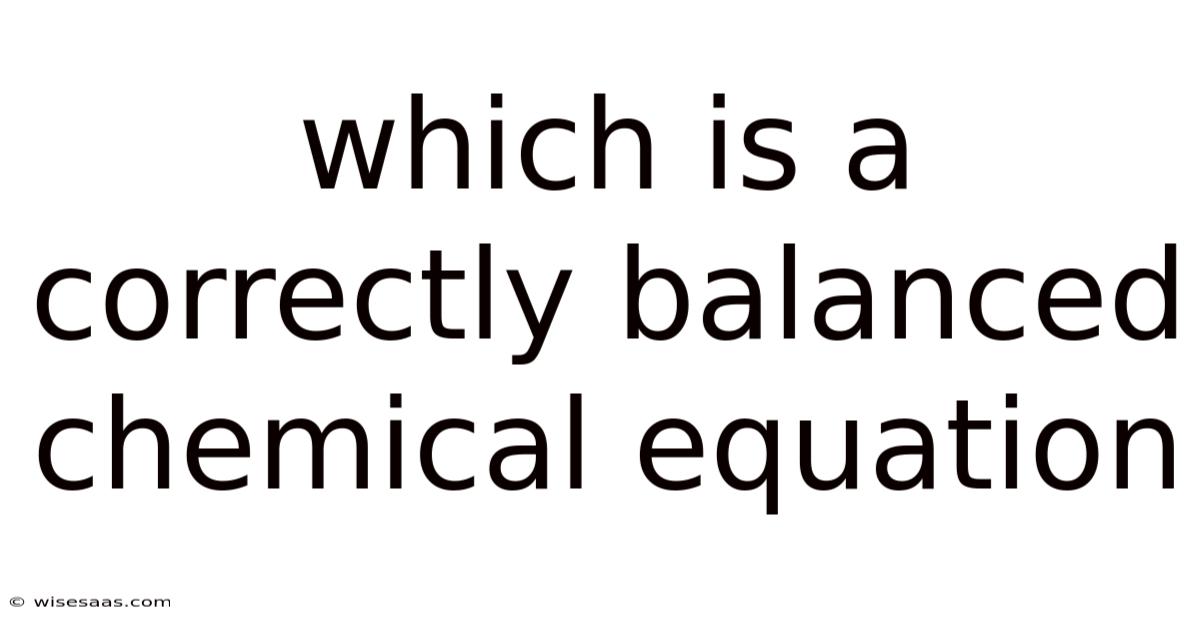Understanding a correctly balanced chemical equation is fundamental in the world of chemistry. That said, whether you're a student, a teacher, or simply someone curious about the building blocks of matter, grasping the concept of balance in chemical equations is essential. This article will guide you through the key points that make a chemical equation balanced, why it matters, and how to identify whether a given equation is correctly balanced.
When we talk about a chemical equation, we are referring to a symbolic representation of a chemical reaction. It shows the reactants on the left side and the products on the right side, along with the changes in the number of atoms for each element. Which means the goal of any chemical equation is to reflect the conservation of mass and charge. So in practice, the total number of atoms for each element must remain the same before and after the reaction.
One of the most important aspects of any chemical equation is its balance. Think about it: a balanced equation ensures that the atoms of every element are equal on both sides of the equation. This is crucial because it reflects the reality of chemical reactions, where every atom must be accounted for. If an equation is not balanced, it indicates an error in the calculation or setup of the reaction.
So, what does it mean for a chemical equation to be balanced? Let's break it down. In a balanced equation, the number of atoms of each element on the reactant side must match the number of atoms of that element on the product side.
The official docs gloss over this. That's a mistake.
2 H₂ + O₂ → 2 H₂O
In this equation, we see that there are 2 hydrogen atoms on the left and 2 hydrogen atoms on the right. Similarly, there are 2 oxygen atoms on the left and 2 oxygen atoms on the right. The equation is balanced because the number of each element is consistent on both sides.
Still, not all chemical equations are immediately obvious as balanced. Think about it: it often requires careful counting and adjustment. The process of balancing involves adjusting coefficients—numbers placed in front of chemical formulas—to make sure the equation is mathematically accurate.
Let’s explore the steps involved in balancing a chemical equation. First, we write the unbalanced equation clearly. Because of that, then, we count the number of atoms for each element on both sides. If the counts differ, we adjust the coefficients accordingly.
Take this case: consider the unbalanced equation:
H₂ + O₂ → H₂O
Here, we have 2 hydrogen atoms and 2 oxygen atoms on the left, but only 1 hydrogen and 2 oxygen atoms on the right. To balance the equation, we need to find a combination where the number of hydrogen and oxygen atoms match on both sides Small thing, real impact..
Counterintuitive, but true.
We can start by balancing the hydrogen atoms. There are 2 hydrogen atoms on the left and 2 on the right. So, we can use a coefficient of 2 in front of H₂:
2 H₂ + O₂ → 2 H₂O
Now, let's count the atoms:
- Left side: 2 × 2 = 4 hydrogen atoms, 2 oxygen atoms.
- Right side: 2 × 2 = 4 hydrogen atoms, 2 oxygen atoms.
Now the equation is balanced. Each element has the same number of atoms on both sides.
Another example might involve more complex molecules. Take the combustion reaction of methane:
CH₄ + 2 O₂ → CO₂ + 2 H₂O
Counting the atoms:
- Left: 1 C, 4 H, 2 O
- Right: 1 C, 2 O, 2 H
We see that the number of carbon atoms matches, and the number of hydrogen and oxygen atoms also match. The equation is balanced.
Balancing isn't always straightforward, especially with molecules that have more than two elements. In such cases, we might need to use a systematic approach, adjusting coefficients one by one until the equation is balanced Simple, but easy to overlook..
It’s important to remember that a balanced chemical equation is not just about numbers—it's about ensuring that the reaction is physically and chemically possible. Take this: if a reaction produces more products than reactants, it may indicate an error in the setup.
One common mistake students make is forgetting to include subscripts in the formulas. Here's a good example: if someone writes:
H₂ + O₂ → H₂O
They might think it's balanced, but actually, it's not. The correct balanced form would be:
2 H₂ + O₂ → 2 H₂O
This ensures that both sides have 2 hydrogen atoms and 2 oxygen atoms The details matter here..
Another point to consider is the use of symbols and notation. In scientific writing, it's crucial to use proper symbols for elements and compounds. Here's one way to look at it: instead of writing "water" as H₂O, we should use the chemical formula H₂O. This not only makes the equation more precise but also helps in identifying imbalances more easily.
Understanding the concept of balance also helps in interpreting the real-world implications of chemical reactions. Consider this: for instance, in industrial processes, balanced equations are essential for calculating the amounts of reactants needed and products formed. This ensures efficiency and safety in chemical manufacturing.
Beyond that, balanced equations are vital in educational settings. Also, teachers often use them to help students grasp the concept of conservation of mass. By working through examples and verifying the balance, students develop a deeper understanding of the principles of chemistry Worth keeping that in mind..
When evaluating a chemical equation for balance, it’s helpful to follow a few guidelines:
- Count the atoms: Start by listing all the atoms in the reactants and products.
- Compare the counts: check that the number of atoms of each element is the same on both sides.
- Adjust coefficients: If necessary, change the numbers in front of chemical formulas to balance the equation.
- Check for errors: Double-check your calculations to avoid mistakes.
If you come across a chemical equation that seems balanced but feels off, don’t hesitate to recheck it. Small errors can lead to significant misunderstandings in more advanced topics Took long enough..
In addition to balancing, it’s important to recognize the importance of scientific notation in chemistry. This notation simplifies the representation of complex formulas and makes it easier to identify imbalances. Take this: instead of writing H₂O₆, you can use the symbol HO₂⁻ or H₂O₂, depending on the context.
Understanding the balance in chemical equations also ties into the broader concept of stoichiometry. This branch of chemistry deals with the quantitative relationships between reactants and products. A balanced equation is the foundation of stoichiometric calculations, which are used in various fields such as environmental science, pharmaceuticals, and engineering.
Worth adding, balanced equations help in predicting reaction outcomes. Worth adding: for example, if a reaction is balanced, you can determine the exact amounts of substances needed or the yield of a product. This is especially useful in laboratory settings where precise measurements are crucial.
In some cases, equations may appear balanced but are not chemically feasible. This happens when the reaction involves the formation of elements that don’t naturally combine in that way. Here's one way to look at it: a reaction that produces a compound with a different element composition than expected is likely unbalanced.
It’s also worth noting that balancing equations is not just a mathematical exercise. It reflects the underlying principles of chemistry, such as the conservation of mass and energy. These principles are fundamental to understanding how matter transforms during chemical reactions And it works..
To reinforce your understanding, let’s look at a few more examples of balanced and unbalanced equations.
Take the reaction between carbon monoxide and oxygen to form carbon dioxide:
CO + O₂ → CO₂
Counting the atoms:
- Left: 1 C, 1 O
- Right: 1 C, 2 O
This is not balanced. To fix it, we need to adjust the coefficients. Multiply the left side by 2 and the right side by 1:
2 CO + O₂ → 2 CO₂
Now, left side: 2 × 1 C = 2 C, 2 × 1 O = 2 O
Right side: 2 × 1 C = 2 C, 2 × 2 O = 4 O
Still not balanced. Let’s try again with 3 CO:
**3 CO + O₂ → 3
Balancing chemical equations is a crucial step in understanding how reactions proceed and ensuring that the laws of conservation are upheld. This process not only clarifies the stoichiometry of a reaction but also reinforces the fundamental principles that govern chemical transformations. When working through these equations, it’s essential to pay close attention to the number of atoms for each element, adjusting coefficients systematically until the equation is fully balanced.
In practice, this skill becomes indispensable as you dig into more complex reactions, whether in industrial processes or academic studies. By mastering this concept, students and professionals alike can predict outcomes with greater accuracy and avoid common pitfalls. It also highlights the importance of precision, as even minor discrepancies can alter the interpretation of a reaction’s feasibility Which is the point..
The official docs gloss over this. That's a mistake.
Beyond balancing, it’s valuable to explore how scientific notation enhances clarity in chemical formulas. So instead of relying solely on full formulas, using standardized symbols can simplify communication and make it easier to spot inconsistencies. This practice is particularly useful when dealing with large molecules or reaction mixtures.
Additionally, recognizing the role of stoichiometry in real-world applications strengthens your grasp of chemistry’s practical relevance. Whether calculating reaction yields or designing experiments, a balanced equation serves as a reliable guide It's one of those things that adds up..
Boiling it down, refining your approach to chemical equations not only sharpens your analytical abilities but also deepens your appreciation for the precision required in scientific inquiry. By consistently applying these principles, you build a stronger foundation in chemistry.
Conclusion: Balancing equations is more than a mathematical task—it’s a vital skill that underpins scientific reasoning and practical applications. Mastering this concept empowers you to approach chemistry with confidence and clarity.