What Is The Percent Composition Of Sulfur In H2so4
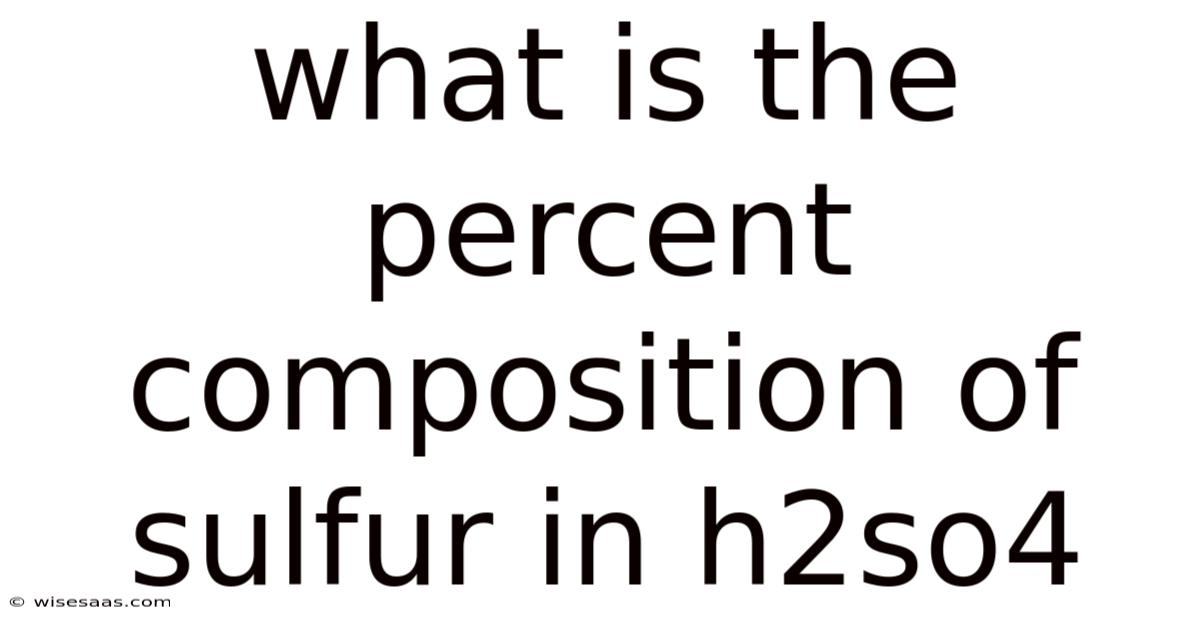
Understanding the percent composition of sulfur in sulfuric acid is essential for anyone delving into chemistry, whether you're a student, educator, or professional. This article will explore the composition of sulfuric acid in detail, breaking down the elements involved and explaining how sulfur fits into this chemical formula. By the end of this guide, you’ll have a clear picture of what sulfur contributes to this vital compound.
Sulfuric acid, often abbreviated as H₂SO₄, is a strong acid widely used in industrial processes, from fertilizer production to chemical synthesis. Its formula clearly shows the presence of sulfur, hydrogen, and oxygen, but what about sulfur specifically? To answer this, we need to look closely at the molecular structure and the percentage that sulfur occupies in the overall formula.
When we examine the chemical formula of sulfuric acid, we see that it consists of two hydrogen atoms, four oxygen atoms, and one sulfur atom. This arrangement is crucial for understanding the percent composition of each element. The percent composition refers to the proportion of each element in the compound relative to the total mass. In this case, we want to focus on sulfur and calculate its share accurately.
To determine the percent composition of sulfur in sulfuric acid, we start by calculating the total number of atoms in the molecule. There are one sulfur atom and four oxygen atoms, along with two hydrogen atoms. The sulfur atom is the key element we’re interested in.
Now, let’s break down the calculation. The formula of sulfuric acid is H₂SO₄, which means there are two hydrogen atoms, four oxygen atoms, and one sulfur atom. To find the percentage of sulfur, we need to divide the number of sulfur atoms by the total number of atoms in the molecule and then multiply by 100.
This process involves a few steps. First, we calculate the total number of atoms: two hydrogen, four oxygen, and one sulfur. That gives us a total of 7 atoms. Now, we focus on sulfur. Since there is only one sulfur atom, we can easily determine its contribution.
Next, we calculate the percentage of sulfur by using the formula:
$ \text{Percent composition of sulfur} = \left( \frac{\text{Number of sulfur atoms}}{\text{Total number of atoms}} \right) \times 100 $
Plugging in the numbers:
$ \text{Percent composition of sulfur} = \left( \frac{1}{7} \right) \times 100 \approx 14.29% $
This means that sulfur makes up approximately 14.29% of the total mass of sulfuric acid. This figure is significant because it highlights the role sulfur plays in the chemical structure and functionality of the acid.
Understanding this percentage is not just about numbers; it’s about grasping how sulfur contributes to the properties of sulfuric acid. Sulfur in this compound is not just a minor player—it is integral to its acidity and reactivity. The sulfur atom forms a central part of the molecule, influencing how sulfuric acid interacts with other substances.
To further clarify, let’s consider the role of sulfur in the chemical behavior of sulfuric acid. Sulfuric acid is a strong acid, and its ability to donate protons (H⁺ ions) is largely due to the presence of sulfur. The sulfur atom can exist in different oxidation states, which affects the acid’s strength and its applications. This adaptability is why sulfuric acid is so valuable in various industries.
It’s important to note that while the percentage of sulfur is approximately 14.29%, the actual mass contribution of sulfur can vary slightly depending on the purity of the acid and the conditions under which it is measured. However, in most standard calculations, this value remains a reliable indicator.
For those who may be curious about the broader context, the importance of sulfur in chemistry cannot be overstated. Sulfur is a fundamental element in many compounds, and its presence in sulfuric acid underscores its significance. This understanding is crucial for students and professionals alike, as it connects chemical concepts to real-world applications.
When exploring the percent composition of sulfur in sulfuric acid, it’s also helpful to compare it with other acids. For instance, in phosphoric acid (H₃PO₄), sulfur is not present at all. In contrast, sulfuric acid clearly includes sulfur, making it a unique compound in the family of carboxylic acids. This distinction is vital for students learning about acid structures and their properties.
Moreover, the calculation of sulfur’s percentage in sulfuric acid is not just an academic exercise. It has practical implications in fields such as environmental science, where sulfuric acid is used in pollution control, and in pharmaceuticals, where its properties are harnessed for specific applications. Understanding this composition helps in making informed decisions in these areas.
In addition to the numerical value, it’s worth considering the significance of this information. Knowing that sulfur constitutes a notable portion of sulfuric acid reinforces the idea that this compound is more than just a chemical formula. It is a product of precise molecular design, where sulfur plays a pivotal role. This insight can inspire learners to appreciate the complexity of chemical substances.
If you’re looking to deepen your understanding of sulfuric acid, it’s essential to recognize how this element contributes to its overall composition. The 14.29% figure serves as a reminder of the balance between different components in this acid. It also highlights the need for accurate measurements and calculations in scientific work.
In conclusion, the percent composition of sulfur in sulfuric acid is a critical aspect of its chemical identity. By understanding this value, we gain a better appreciation for the role sulfur plays in this essential compound. This knowledge not only aids in academic studies but also enhances practical applications in various industries. Whether you’re studying chemistry or exploring its uses, this information is a valuable piece of the puzzle.
Remember, the details matter. The presence of sulfur in sulfuric acid is not just a statistic—it’s a foundation for its functionality and utility. As you continue your journey through chemistry, keep this information in mind, and let it guide your curiosity and learning.
Continuing from theestablished context, it's crucial to recognize that the 14.29% sulfur composition is not merely a static figure; it actively shapes the acid's defining characteristics. This specific proportion underpins sulfuric acid's formidable strong acidic strength, a property directly harnessed in its primary industrial role: concentrating and dehydrating. The high concentration of sulfur, bound within the sulfate ion (SO₄²⁻), provides the molecular framework that allows sulfuric acid to act as a potent dehydrating agent, stripping water molecules from other substances with remarkable efficiency. This capability is fundamental to processes like the production of esters, the drying of gases, and the synthesis of various chemicals where moisture control is critical.
Furthermore, this elemental balance influences the acid's behavior in complex mixtures. For instance, when sulfuric acid interacts with metals or other compounds, the sulfur content dictates the reaction pathways and products. The presence of sulfur also contributes to the acid's high boiling point (decomposing before boiling) and its high density in concentrated forms, properties that are directly tied to the mass of the sulfur atom relative to the entire molecule. Understanding this composition allows chemists to predict and manipulate these behaviors, ensuring safe handling and effective application in laboratories and factories.
Beyond its intrinsic chemical properties, the 14.29% sulfur figure carries significant weight in environmental chemistry. Sulfuric acid is a major component of acid rain, formed when sulfur dioxide (SO₂) emissions react with atmospheric water vapor and oxygen. The percentage composition of sulfur in the acid formed from these reactions is a direct indicator of the sulfur load introduced into the environment. Accurate knowledge of this percentage is vital for modeling atmospheric chemistry, predicting the impact on ecosystems and infrastructure, and developing effective strategies for pollution control and mitigation. It transforms a simple chemical formula into a key parameter for understanding and addressing a global environmental challenge.
In the realm of materials science and engineering, the precise sulfur content influences the performance of sulfuric acid in specific applications. For example, in the lead-acid battery, the sulfuric acid electrolyte's composition is critical for maintaining the correct ion concentration and conductivity, directly impacting battery life and efficiency. The sulfur's presence is not incidental; it's engineered into the system's functionality. Similarly, in the production of fertilizers like superphosphate, the sulfuric acid's sulfur content is a primary nutrient source, and its concentration affects the solubility and availability of phosphorus to plants.
Ultimately, the 14.29% sulfur in sulfuric acid serves as a powerful reminder of the profound link between elemental composition and macroscopic properties. It underscores that chemistry is not just about abstract symbols, but about understanding how the fundamental building blocks – like sulfur – dictate the behavior, utility, and impact of the substances that shape our world. This knowledge empowers scientists, engineers, and policymakers to innovate, solve problems, and make informed decisions across diverse fields, from developing cleaner industrial processes to designing more efficient chemical products and mitigating environmental damage. The significance of sulfur's role, quantified precisely, is a cornerstone of chemical literacy and practical application.
Latest Posts
Latest Posts
-
People Who Work In High Stress Professions
Mar 28, 2026
-
Using Social Media To Support Activities Such As Producing
Mar 28, 2026
-
Fostering A Leadership Environment Involves All Of The Following Except
Mar 28, 2026
-
Which Sentence Correctly Uses A Pronoun With A Clear Antecedent
Mar 28, 2026
-
The Victorian Era Of Literature Occurred Between
Mar 28, 2026