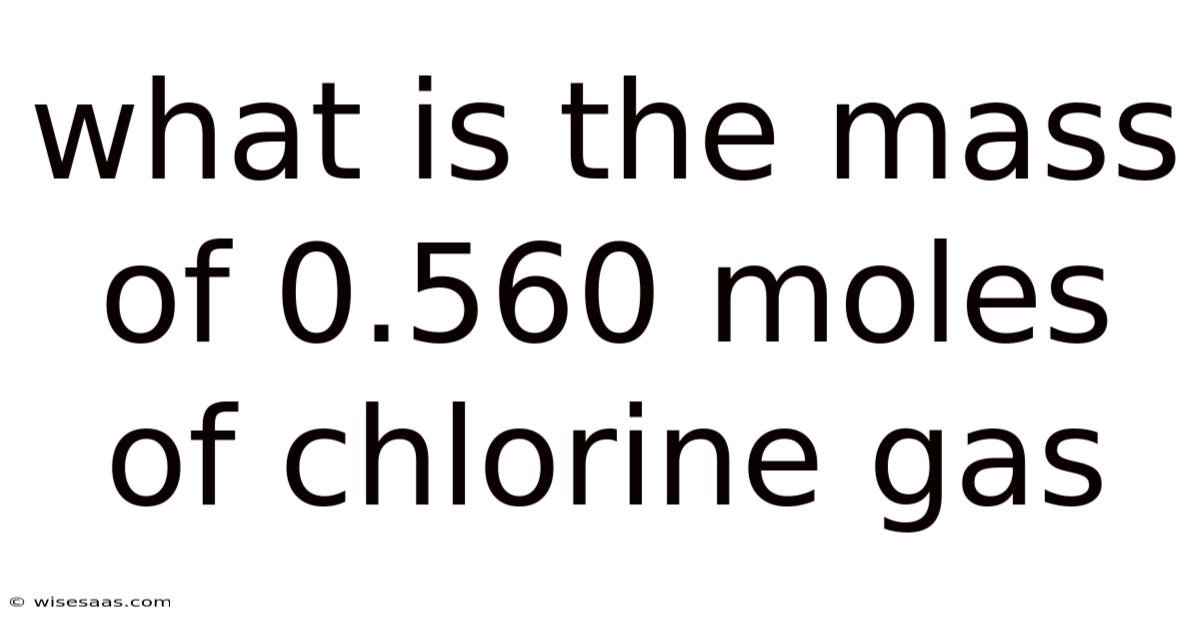What Is the Mass of 0.560 Moles of Chlorine Gas and How to Calculate It Using Molar Mass
Understanding the relationship between moles, mass, and molar mass is fundamental in chemistry, especially when dealing with gases such as chlorine gas. And if you have ever wondered what is the mass of 0. 560 moles of chlorine gas, you are engaging with a core concept in stoichiometry. In real terms, this calculation requires knowledge of the molar mass of chlorine gas, the distinction between chlorine atoms and chlorine molecules, and the proper application of the mole concept. This article will walk you through the steps, explain the scientific reasoning, and provide clarity so you can confidently solve similar problems.
Introduction to the Mole Concept and Chlorine Gas
The mole is a unit that measures the amount of substance, much like a dozen measures quantity. One mole of any substance contains Avogadro's number of particles, which is approximately 6.022 × 10²³ entities. When dealing with gases, chemists often need to convert between the number of moles and the mass in grams. Chlorine gas, which exists as diatomic molecules (Cl₂), is a common example where this conversion is essential Not complicated — just consistent..
To find the mass of 0.560 moles of chlorine gas, you must first determine the molar mass of Cl₂. The molar mass is the mass of one mole of a substance and is expressed in grams per mole (g/mol). It is numerically equal to the atomic mass of the element as found on the periodic table, but for a molecule, you must sum the atomic masses of all atoms in the molecule.
Steps to Calculate the Mass of 0.560 Moles of Chlorine Gas
The calculation can be broken down into clear, logical steps:
-
Identify the chemical formula: Chlorine gas is diatomic, meaning it consists of two chlorine atoms bonded together. Its formula is Cl₂.
-
Find the atomic mass of chlorine: Look up the atomic mass of chlorine on the periodic table. It is approximately 35.45 atomic mass units (amu) No workaround needed..
-
Calculate the molar mass of Cl₂: Since one molecule of chlorine gas contains two chlorine atoms, multiply the atomic mass by 2 Most people skip this — try not to..
- Molar mass of Cl₂ = 2 × 35.45 g/mol = 70.90 g/mol.
-
Use the mole-to-mass conversion formula: The mass of a substance in grams is equal to the number of moles multiplied by the molar mass.
- Mass = moles × molar mass
-
Substitute the values: You are given 0.560 moles of chlorine gas.
- Mass = 0.560 mol × 70.90 g/mol
-
Perform the calculation: Multiply 0.560 by 70.90 And that's really what it comes down to..
- 0.560 × 70.90 = 39.704
-
Apply significant figures: The given value, 0.560, has three significant figures. So, the final answer should also be reported with three significant figures.
- Mass ≈ 39.7 grams
Thus, the mass of 0.560 moles of chlorine gas is approximately 39.7 grams Simple, but easy to overlook..
Scientific Explanation: Why Molar Mass Matters
The concept of molar mass serves as a bridge between the microscopic world of atoms and molecules and the macroscopic world of grams and liters. Because atoms and molecules are incredibly small, counting them individually is impractical. Instead, chemists use the mole as a counting unit, and molar mass allows for the conversion between this count and measurable mass.
It sounds simple, but the gap is usually here Easy to understand, harder to ignore..
For chlorine gas, the diatomic nature is crucial. If you mistakenly used the atomic mass of chlorine (35.Which means 45 g/mol) instead of the molecular mass (70. Also, 90 g/mol), your answer would be off by a factor of two. This highlights the importance of understanding molecular structure. The molar mass of a compound reflects the total mass of all atoms in one mole of that compound Worth knowing..
Additionally, this calculation assumes pure chlorine gas at standard conditions. Think about it: while pressure and temperature can affect the volume of a gas, they do not affect the molar mass or the mass of a specific number of moles. So, the mass remains constant regardless of the physical state or conditions, as long as the number of moles does not change Still holds up..
Common Mistakes and How to Avoid Them
When solving problems like "what is the mass of 0.560 moles of chlorine gas," students often encounter pitfalls. One common error is confusing chlorine atoms (Cl) with chlorine gas (Cl₂). Always verify whether the substance is atomic or molecular. For gases like chlorine, oxygen, nitrogen, and hydrogen, the diatomic form is standard Small thing, real impact..
Another mistake is neglecting significant figures. In scientific calculations, significant figures convey the precision of a measurement. Reporting the answer as 39.Since the input value (0.560) has three significant figures, the answer must reflect that precision. 704 grams implies a level of accuracy that is not justified Small thing, real impact. Practical, not theoretical..
Finally, some learners forget to multiply the atomic mass by the subscript in the chemical formula. Remember, the subscript indicates the number of atoms in the molecule. For Cl₂, the "2" is essential for calculating the correct molar mass.
Frequently Asked Questions
Q: What is the molar mass of chlorine gas?
A: The molar mass of chlorine gas (Cl₂) is approximately 70.90 g/mol. This value is derived by adding the atomic masses of two chlorine atoms, each contributing about 35.45 g/mol.
Q: Why is chlorine gas diatomic?
A: Chlorine is a halogen and exists as a diatomic molecule in its natural gaseous state. In plain terms, two chlorine atoms bond together to form Cl₂, which is more stable than individual chlorine atoms Worth keeping that in mind..
Q: How does the number of significant figures affect the answer?
A: Significant figures make sure the calculated mass reflects the precision of the input data. Since 0.560 has three significant figures, the final mass should be reported as 39.7 grams, not 39.704 grams Nothing fancy..
Q: Can this method be applied to other gases?
A: Yes, the same approach works for any gas. You need to determine the correct molecular formula, calculate the molar mass, and then multiply by the number of moles. As an example, oxygen gas (O₂) would use a molar mass of approximately 32.00 g/mol.
Q: Does the physical state of chlorine affect the calculation?
A: No, the mass of a specific number of moles is independent of the physical state. Whether chlorine is a gas, liquid, or solid, the molar mass remains the same Practical, not theoretical..
Conclusion and Practical Application
Calculating the mass of 0.560 moles of chlorine gas is a straightforward application of the mole concept and molar mass. By recognizing that chlorine gas is diatomic and using the correct molar mass of 70.90 g/mol, you can determine that the mass is approximately 39.Now, 7 grams. This skill is not only important for academic exercises but also for real-world applications in chemistry, such as preparing solutions, conducting experiments, and understanding chemical reactions Not complicated — just consistent..
Mastering these calculations builds a strong foundation for more advanced topics in chemistry, including stoichiometry, gas laws, and chemical equilibrium. Whenever you encounter a similar problem, remember to identify the molecular formula, find the molar mass, and apply the mole-to-mass conversion. With practice, these steps will become second nature, allowing you to tackle more complex chemical calculations with confidence Nothing fancy..
Conclusion and Practical Application
The ability to accurately calculate the mass of chlorine gas is not only a fundamental skill in chemistry education but also a critical tool in various fields, including pharmaceuticals, materials science, and environmental chemistry. Understanding the molar mass of chlorine gas and its diatomic nature is essential for correctly interpreting chemical equations and predicting reaction outcomes.
In practical scenarios, such as laboratory work or industrial processes, precise knowledge of molar masses is crucial for ensuring safety, efficiency, and accuracy. Here's a good example: when preparing a chlorine-based disinfectant, knowing the exact mass of chlorine gas required ensures that the solution is effective and safe Easy to understand, harder to ignore..
On top of that, this calculation is a stepping stone to more complex problems, such as determining the empirical and molecular formulas of compounds, analyzing chemical compositions, and understanding the behavior of gases under different conditions. By mastering these basics, students and professionals alike can confidently approach more advanced topics and real-world applications.
At the end of the day, the calculation of the mass of 0.Consider this: this exercise reinforces the importance of attention to detail, such as recognizing diatomic molecules and applying significant figures correctly. 7 grams. 90 g/mol, results in 39.560 moles of chlorine gas, with a molar mass of approximately 70.These principles are not just academic; they are vital for anyone engaged in chemical sciences, ensuring that theoretical knowledge translates into practical, effective solutions Nothing fancy..