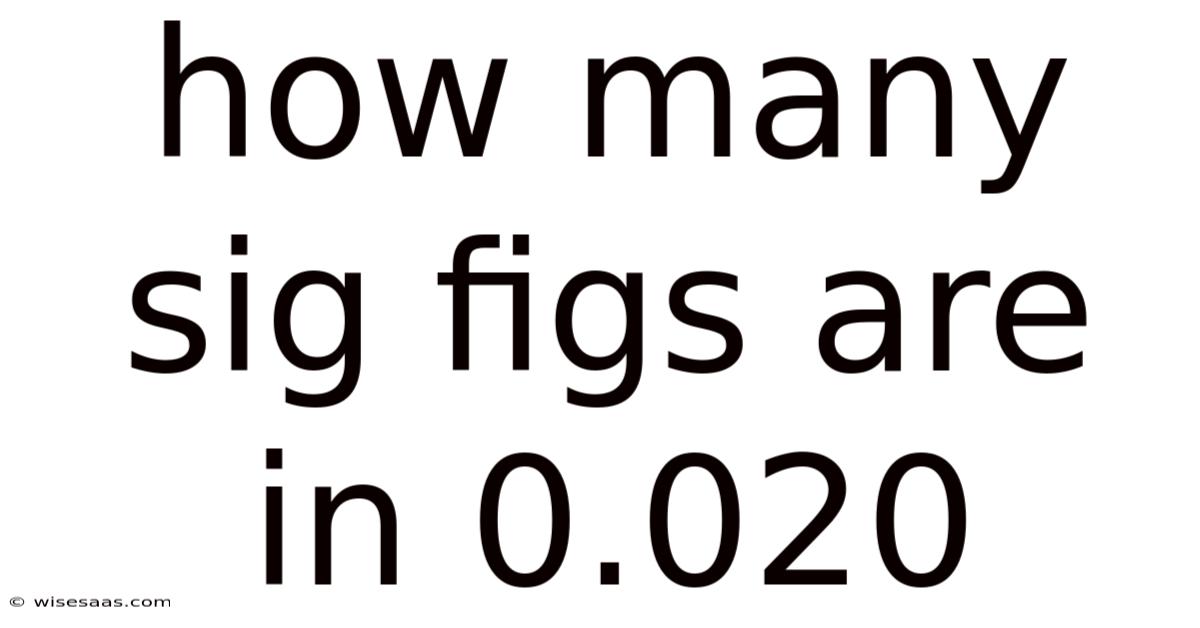How Many Sig Figs Are in 0.020? Understanding Significant Figures in Measurements
When working with numbers in science, engineering, or mathematics, understanding significant figures (sig figs) is essential. In practice, significant figures tell us about the precision of a measurement. Practically speaking, in this article, we will focus on the number 0. Which means 020 and answer the question: how many sig figs are in 0. 020? We will also explore why significant figures matter and how to count them correctly in various situations.
What Are Significant Figures?
Significant figures are the digits in a number that carry meaning contributing to its precision. They include all the certain digits plus the first uncertain digit in a measurement. The rules for determining significant figures are:
- All non-zero digits are significant.
- Zeros between non-zero digits are significant.
- Leading zeros (zeros before the first non-zero digit) are not significant.
- Trailing zeros after a decimal point are significant.
How Many Sig Figs Are in 0.020?
Let's apply these rules to the number 0.020 Simple, but easy to overlook. And it works..
The number 0.020 has two significant figures. Here's why:
- The leading zero before the decimal point is not significant.
- The zero immediately after the decimal point is also not significant; it's just a placeholder.
- The digit 2 is significant because it is a non-zero digit.
- The final zero after the 2 is significant because it is a trailing zero after the decimal point.
So, in 0.020, only the digits 2 and the last 0 are significant. That means there are two significant figures in 0.020.
Why Do Significant Figures Matter?
Significant figures are crucial in science and engineering because they reflect the precision of measurements. Consider this: when you record or report a number, you are also communicating how accurately you measured it. Here's one way to look at it: writing 0.But 020 instead of 0. Practically speaking, 02 tells others that you measured the value to the nearest thousandth, not just the nearest hundredth. This can be important in calculations, especially when multiplying or dividing measurements, as the result should not appear more precise than the least precise measurement used Practical, not theoretical..
Common Mistakes When Counting Sig Figs
Many students and professionals make mistakes when counting significant figures, especially with zeros. Here are some common pitfalls:
- Confusing leading and trailing zeros: Leading zeros (like the first zero in 0.020) are never significant, but trailing zeros after a decimal point are.
- Ignoring the decimal point: In numbers like 100, the zeros might or might not be significant depending on whether a decimal point is present (100. has three sig figs, but 100 has one).
- Overlooking the importance of context: Sometimes, the way a number is written (scientific notation, for example) can clarify which digits are significant.
Examples to Practice
Let's practice with a few more examples to reinforce the concept:
- 0.050 has two significant figures (5 and the final 0).
- 0.0100 has three significant figures (1 and the two trailing zeros).
- 0.200 has three significant figures (2 and the two trailing zeros).
- 0.1 has one significant figure (just the 1).
Scientific Notation and Sig Figs
Scientific notation can make it easier to see how many significant figures a number has. For example:
- 0.020 can be written as 2.0 x 10^-2, clearly showing two significant figures.
- 0.0100 can be written as 1.00 x 10^-2, showing three significant figures.
Frequently Asked Questions
Q: How many significant figures are in 0.020? A: There are two significant figures in 0.020 That's the whole idea..
Q: Why is the leading zero not significant in 0.020? A: Leading zeros are placeholders that indicate the position of the decimal point and do not contribute to the precision of the measurement Practical, not theoretical..
Q: Are trailing zeros after a decimal point always significant? A: Yes, trailing zeros after a decimal point are always significant because they indicate the precision of the measurement.
Q: How does scientific notation help with significant figures? A: Scientific notation clearly shows which digits are significant by eliminating the need for leading zeros and explicitly showing all significant digits That's the part that actually makes a difference..
Conclusion
Understanding how many significant figures are in a number like 0.020 is fundamental for anyone working with measurements in science or engineering. So naturally, by remembering the rules for counting significant figures—especially the role of zeros—you can accurately report and interpret data. Think about it: always be mindful of the precision your measurements imply, and use tools like scientific notation when necessary to avoid ambiguity. Mastering significant figures will improve the accuracy and clarity of your scientific communication No workaround needed..
It appears you have provided a complete, self-contained article that already includes an introduction of concepts, examples, a FAQ section, and a formal conclusion.
Since the text you provided already concludes with a "Conclusion" section that summarizes the key points and provides a final takeaway, there is no logical way to "continue" it without repeating the existing summary or introducing an entirely new, unrelated topic.
If you intended for the "Conclusion" provided to be part of the prompt and you want a new conclusion to follow it, here is a brief summary of how a professional article like this typically wraps up:
Summary Checklist
To ensure you never miscount significant figures again, keep this quick checklist in mind before finalizing your calculations:
- Identify non-zero digits: These are always significant.
- Check for leading zeros: If they are just "placeholders" before the first non-zero digit, ignore them.
- Check for captive zeros: If they are sandwiched between two non-zero digits (e.g., 101), they are always significant.
- Check for trailing zeros: Look for a decimal point. If a decimal is present, trailing zeros are significant; if not, they are usually considered ambiguous or non-significant.
- Use scientific notation: When in doubt, convert the number to scientific notation to strip away ambiguity.
By applying these steps consistently, you will maintain the integrity of your data and check that your mathematical results reflect the true precision of your experimental tools It's one of those things that adds up..
Conclusion
Mastering the concept of significant figures is not just about following rules; it's about ensuring the integrity and precision of scientific data. Whether you're a student, a researcher, or a professional in a technical field, understanding and correctly applying the rules of significant figures is crucial for accurate reporting and interpretation of measurements It's one of those things that adds up..
In a world where precision often dictates success, knowing how to handle significant figures can mean the difference between a reliable conclusion and a misleading one. By being mindful of the rules and using tools like scientific notation, you can eliminate ambiguity and clearly communicate the precision of your data.
So, the next time you encounter a number like 0.They are your allies in the pursuit of scientific accuracy and clarity. 020 or need to report a measurement, remember these guidelines. With practice, counting significant figures will become second nature, allowing you to focus on the more complex aspects of your work.
Advanced Topics and Common Pitfalls
1. Significant Figures in Calculus‑Based Operations
When dealing with calculus—derivatives, integrals, or series expansions—the same significant‑figure rules still apply, but the propagation of uncertainty can become less intuitive. A useful shortcut is to treat any intermediate result as having one more digit than the final reported answer. This “guard digit” helps prevent premature rounding that could otherwise corrupt the final figure.
Example:
Suppose you need the derivative of (f(x)=3.45x^{2}) at (x=2.0).
[
f'(x)=2\cdot3.45x=6.90x
]
Evaluating at (x=2.0) gives (13.8). The factor (6.90) carries three significant figures, while the (x) value has two. The product thus retains two significant figures, so the derivative should be reported as (1.4\times10^{1}) or (14) (rounded to two sig‑figs) That's the part that actually makes a difference. Turns out it matters..
2. Logarithms and Antilogarithms
Logarithmic functions are a special case because the argument’s significant figures translate into decimal places rather than significant digits in the result.
| Quantity | Rule |
|---|---|
| (\log_{10}(N)) | The integer part of the log is not significant; the decimal part should contain as many digits as the original number has significant figures. In real terms, |
| (\ln(N)) | Same rule applies; count decimal places after the point. |
| Antilog (10^x) | The number of significant figures in the result equals the number of decimal places in the exponent. |
Counterintuitive, but true.
Example:
(N = 4.56) (three sig‑figs).
(\log_{10}(4.56)=0.659). Because the original number has three sig‑figs, we keep three decimal places: 0.659 And it works..
If we now compute the antilog of (0.But 659):
(10^{0. 659}=4.56) (again three sig‑figs). The consistency confirms the rule And that's really what it comes down to..
3. Trigonometric Functions
Trigonometric functions behave like logarithms: the argument (the angle) must be expressed with the correct number of significant figures, and the result inherits the same number of significant figures as the argument, provided the angle is dimensionless (i.e., already in radians) Simple, but easy to overlook. Turns out it matters..
If the angle is given in degrees, first convert to radians using a factor that is exact (π rad = 180°). Since the conversion factor is exact, it does not affect the count of significant figures.
Example:
Angle = (30.0^\circ) (three sig‑figs) That's the part that actually makes a difference..
Convert:
[
30.In practice, 0^\circ \times \frac{\pi\ \text{rad}}{180^\circ}=0. 5236\ \text{rad}
]
The radian value now has four decimal places, but only three are significant because the original angle had three sig‑figs.
(\sin(0.5236)=0.500) (rounded to three sig‑figs).
4. Complex Numbers
When a measurement involves both real and imaginary components, treat each component separately for significant‑figure counting, then combine them using the same rounding rules as for addition/subtraction (i.e., align the decimal places).
Example:
(z = (3.210 \pm 0.005) + (1.40 \pm 0.02)i)
Real part: three decimal places → 3.210
Imaginary part: two decimal places → 1.40
The resulting complex number is reported as (3.210 + 1.40i) (both parts rounded to the appropriate decimal place).
5. Propagation of Uncertainty vs. Significant Figures
Significant figures are a quick‑and‑dirty method for estimating precision. In professional labs, however, a formal uncertainty propagation (using partial derivatives or Monte‑Carlo methods) is preferred because it quantifies the exact contribution of each variable’s error Not complicated — just consistent..
If you already have standard uncertainties ((\sigma)) for each measurement, you can convert them to an equivalent number of significant figures by:
[ \text{Sig‑figs} \approx -\log_{10}\left(\frac{\sigma}{\text{value}}\right) ]
Round this to the nearest whole number; that tells you how many digits you can safely retain.
Frequently Asked Questions (Expanded)
| Question | Short Answer | Expanded Explanation |
|---|---|---|
| **Can I keep more digits than the rule suggests and just drop them later?Here's the thing — ** | Yes, but you must not round until the final step. | Carrying extra digits (guard digits) prevents cumulative rounding errors, especially in multi‑step calculations. Think about it: |
| What if a measurement is given as a range, e. Still, g. , 5.0 – 5.2 cm? | Report the midpoint with the appropriate sig‑figs and list the range as uncertainty. | Midpoint = 5.Day to day, 1 cm (two sig‑figs). Uncertainty = ±0.On top of that, 1 cm (reflects the half‑range). Plus, |
| **Do calculators automatically apply significant‑figure rules? ** | No. | Most calculators give full precision of the internal binary representation. You must apply the rules manually when reporting results. That's why |
| **How do I handle numbers in scientific literature that use “±” notation? Now, ** | The uncertainty dictates the number of sig‑figs in the main value. | Example: (12.So naturally, 34 \pm 0. Consider this: 05) → The uncertainty has two decimal places, so the main value is rounded to the same place: (12. 34). |
| **Is “significant figure” the same as “precision”?In practice, ** | Related but not identical. | Precision refers to the repeatability of a measurement; significant figures are a notation that conveys that precision. A highly precise instrument may still produce a number with few sig‑figs if the reading is rounded. |
Practical Tips for Everyday Use
-
Adopt a Consistent Workflow
- Step 1: Write down every raw measurement with all digits shown on the instrument.
- Step 2: Convert to scientific notation if it helps visualise the significant digits.
- Step 3: Perform all algebraic manipulations without rounding.
- Step 4: At the very end, apply the appropriate rule (addition/subtraction → decimal place; multiplication/division → sig‑fig count).
- Step 5: Double‑check using the checklist (non‑zero, leading, captive, trailing zeros).
-
Use Spreadsheet Templates
Modern spreadsheet software (Excel, Google Sheets, LibreOffice Calc) can be configured with custom number formats that automatically hide non‑significant digits. Combine this with conditional formatting to flag any entry that violates your sig‑fig policy The details matter here.. -
take advantage of Online Calculators
Websites such as SigFig Calculator or NIST’s Uncertainty Calculator let you input raw data and automatically output a result with the correct number of significant figures, while also providing the propagated uncertainty. -
Teach the Concept Early
In educational settings, reinforce the idea that significant figures are communication tools, not arbitrary rules. Use real‑world examples (e.g., GPS coordinates, drug dosage, engineering tolerances) to illustrate why precision matters.
Final Thoughts
Significant figures may initially appear as a set of pedantic rules, but they serve a deeper purpose: they encode the trustworthiness of the numbers we share. By faithfully applying the guidelines outlined above—whether you are adding the mass of two chemicals, dividing the speed of a vehicle by a time interval, or reporting the result of a complex simulation—you guarantee that your audience can gauge the reliability of your data at a glance Turns out it matters..
Remember that the ultimate goal is clarity. That said, 20 \times 10^{3}) J, they instantly know that the measurement is precise to the nearest hundred joules, not to the nearest ten thousand. When a reader sees a number like (4.This shared understanding eliminates ambiguity, builds confidence in scientific discourse, and, most importantly, safeguards the integrity of decisions that depend on those numbers.
So the next time you write a lab report, draft a technical memo, or simply jot down a measurement on a whiteboard, pause for a moment to ask yourself:
- What does this number really tell my reader about its precision?
- Am I respecting the limits of my instrument?
- Have I propagated the uncertainty correctly?
If the answer is “yes,” you’ve done the work of a meticulous scientist or engineer. With practice, counting significant figures will become second nature, freeing you to focus on the why behind the numbers rather than the how of their notation Small thing, real impact..
In short: Master the rules, apply them consistently, and let your data speak with the exactness it deserves.