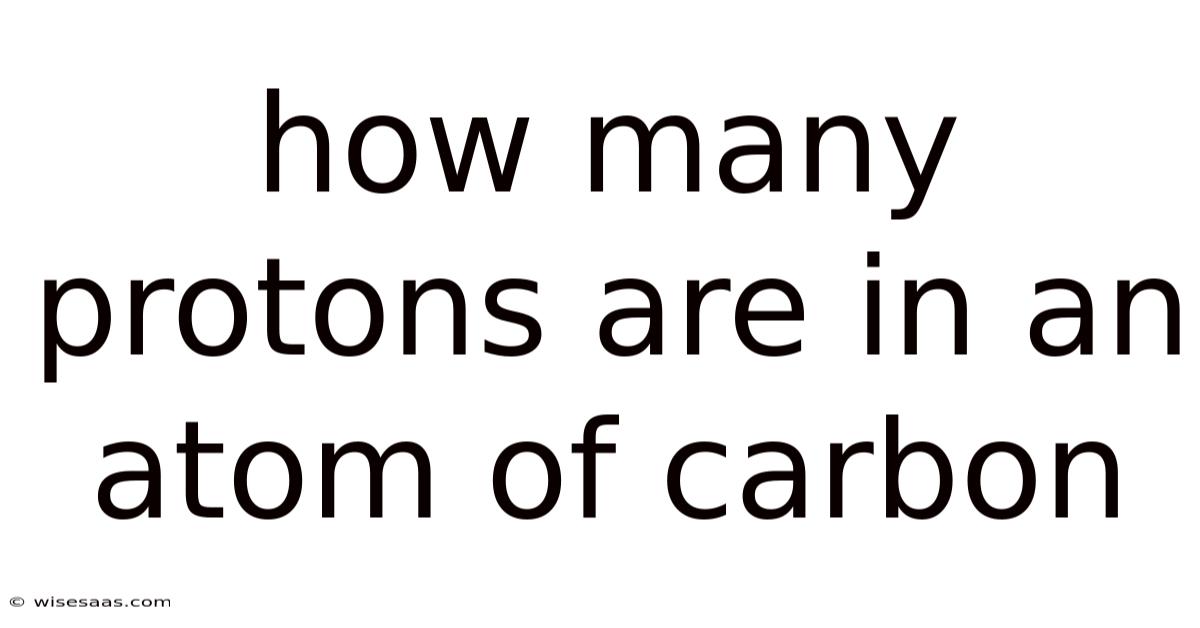How Many Protons Are in an Atom of Carbon?
Understanding the fundamental building blocks of matter is essential in the study of chemistry and physics. At the heart of this exploration lies the atom, the smallest unit of an element that retains the chemical properties of that element. That's why among the myriad elements that make up our universe, carbon is one of the most intriguing and abundant. Day to day, in this article, we will get into the question: *How many protons are in an atom of carbon? * This seemingly straightforward inquiry opens a gateway to understanding atomic structure and the periodic table No workaround needed..
The Basics of Atomic Structure
Before we answer the question, let's briefly review the basic components of an atom. Atoms are composed of three types of subatomic particles: protons, neutrons, and electrons. Protons and neutrons reside in the atom's nucleus, which is the central core of the atom. Electrons, on the other hand, exist in a cloud of space around the nucleus. The number of protons in an atom's nucleus determines the element's identity on the periodic table.
Protons and Atomic Number
The number of protons in an atom's nucleus is known as its atomic number. This number is unique to each element and defines the element's position on the periodic table. Here's a good example: hydrogen has an atomic number of 1, meaning it has one proton in its nucleus. Similarly, helium has an atomic number of 2, indicating it has two protons.
Carbon, with an atomic number of 6, has six protons in its nucleus. This atomic number places carbon in the second period and the fourth group of the periodic table, just below nitrogen and above boron. The six protons give carbon its unique chemical properties and position in the periodic table.
Isotopes and the Proton Count
While the number of protons in an atom is fixed and defines the element, the number of neutrons can vary, creating different isotopes of the same element. Isotopes have the same number of protons but a different number of neutrons, which affects the atom's mass but not its chemical properties Worth keeping that in mind..
Carbon, for example, has several isotopes, including carbon-12, carbon-13, and carbon-14. Now, carbon-12, the most abundant isotope, has six protons and six neutrons, giving it a mass number of 12. Which means carbon-13 has six protons and seven neutrons, resulting in a mass number of 13. Even so, carbon-14, less common and radioactive, has six protons and eight neutrons, leading to a mass number of 14. Despite these differences in neutron count, all carbon isotopes have six protons and thus belong to the same element.
The Role of Protons in Chemical Bonding
The six protons in a carbon atom play a crucial role in its ability to form chemical bonds. Protons, being positively charged, attract electrons, which are negatively charged. This attraction is the basis of chemical bonding, where atoms combine to form molecules Easy to understand, harder to ignore..
Carbon's six protons mean it has six electrons, which it can share or transfer to form bonds with other atoms. This versatility allows carbon to form a vast array of organic compounds, making it the cornerstone of life as we know it. Carbon can form single, double, and triple bonds with other atoms, including other carbon atoms, which leads to the complexity and diversity of organic molecules.
Carbon in the Periodic Table
Carbon's position on the periodic table is a testament to its importance in the universe. As the second lightest element after hydrogen, carbon is abundant in the cosmos. It is a key component of stars, planets, and even life itself. Carbon's six protons and its ability to form stable bonds with other elements make it a fundamental building block of organic chemistry The details matter here..
No fluff here — just what actually works.
Conclusion
At the end of the day, an atom of carbon contains six protons, as defined by its atomic number. This number is crucial in determining the element's identity and its position on the periodic table. While the number of protons remains constant for a given element, the number of neutrons can vary, leading to different isotopes. The six protons in carbon also enable its unique ability to form a wide range of chemical bonds, making it essential to organic chemistry and life itself Nothing fancy..
Understanding the number of protons in an atom is just the beginning of exploring the fascinating world of atomic structure and the periodic table. As we continue to uncover more about the elements and their properties, we gain deeper insights into the nature of matter and the universe around us Not complicated — just consistent..
Carbon's Versatility: A Foundation for Life
Beyond its fundamental properties, carbon's chemical behavior is remarkably versatile, largely stemming from its ability to form long chains and complex ring structures. This characteristic is driven by the stable covalent bonds it can create with itself and other elements like hydrogen, oxygen, and nitrogen. These bonds are strong enough to form the diverse molecular structures that constitute proteins, carbohydrates, lipids, and nucleic acids – the essential macromolecules of life. The ability to form such involved and stable structures is unparalleled among the elements, making carbon uniquely suited to serve as the backbone of organic chemistry And that's really what it comes down to..
Beyond that, carbon exists in various allotropes, different structural forms of the same element. Diamond, with its rigid tetrahedral structure, showcases carbon's hardness and exceptional thermal conductivity. Because of that, graphite, in contrast, possesses a layered structure that allows for electrical conductivity and its use as a lubricant. Also, fullerenes, spherical carbon molecules, and carbon nanotubes represent even more exotic allotropes with unique and potentially revolutionary applications in materials science and nanotechnology. These diverse forms further highlight the adaptability of carbon and its profound impact on the physical world But it adds up..
The prevalence of carbon-based molecules in the universe is not merely a coincidence; it's a consequence of the element's unique combination of properties. Its abundance, coupled with its ability to form stable and complex bonds, makes it the ideal building block for the complex molecules necessary for life and the diverse materials we rely on. From the simplest organic compounds to the most complex biological systems, carbon’s influence is undeniable.
To wrap this up, the story of carbon, from its atomic structure to its diverse allotropes and crucial role in life, is a testament to the elegance and complexity of the natural world. The six protons defining its identity access a cascade of chemical possibilities, leading to the incredible diversity and functionality we observe around us. Understanding carbon is not just understanding an element; it's understanding the foundation of organic chemistry, the basis of life, and a key to unlocking the secrets of the universe Small thing, real impact. Which is the point..
Carbon in the Modern World: From Industry to Innovation
While carbon’s natural roles are extraordinary, humanity has harnessed its properties to drive technological progress on a massive scale. In the energy sector, carbon compounds such as hydrocarbons—methane, gasoline, diesel—have powered the industrial revolution and continue to dominate global transportation. Also, the combustion of these fuels releases energy stored in carbon‑hydrogen bonds, but it also produces carbon dioxide (CO₂), a greenhouse gas that contributes to climate change. This duality underscores the importance of developing carbon‑neutral or carbon‑negative technologies, such as carbon capture and storage (CCS), bio‑derived fuels, and synthetic hydrocarbons produced from renewable electricity.
In the realm of materials science, carbon’s allotropes have opened doors to products once thought impossible. Now, graphene, a single layer of carbon atoms arranged in a hexagonal lattice, exhibits extraordinary tensile strength, electrical conductivity, and optical transparency. Its potential applications range from flexible touchscreens and high‑frequency transistors to ultrafast charging batteries and desalination membranes. Meanwhile, carbon nanotubes—cylindrical sheets of graphene—offer unparalleled strength‑to‑weight ratios, making them ideal candidates for lightweight composites in aerospace, sports equipment, and next‑generation prosthetics.
Carbon’s role extends even further into the digital age. The semiconductor industry relies heavily on carbon‑based materials for doping silicon wafers, creating conductive pathways, and developing resistive memory devices. In quantum computing, researchers are exploring carbon‑vacancy centers in diamond as stable qubits that can operate at room temperature, promising a scalable route toward practical quantum processors Took long enough..
The Environmental Dimension: Carbon Cycling and Climate
Understanding carbon’s chemical versatility also requires an appreciation of the global carbon cycle—a set of interconnected processes that move carbon among the atmosphere, hydrosphere, lithosphere, and biosphere. Photosynthesis captures atmospheric CO₂, converting it into organic matter that fuels ecosystems. Respiration, decomposition, and combustion return carbon to the atmosphere, completing the loop. Human activities have perturbed this balance, accelerating the accumulation of CO₂ and other greenhouse gases.
Mitigating these impacts hinges on both reducing carbon emissions and enhancing natural carbon sinks. Reforestation and afforestation increase the biosphere’s capacity to sequester carbon, while soil management practices—such as no‑till farming and biochar addition—lock carbon into the ground for centuries. On the technological front, direct air capture (DAC) systems chemically bind CO₂ from ambient air, allowing it to be stored underground or repurposed into fuels and building materials.
Emerging Frontiers: Carbon in Synthetic Biology and Space Exploration
The intersection of carbon chemistry with synthetic biology is yielding novel pathways to produce valuable compounds sustainably. In practice, engineered microbes can convert inexpensive feedstocks like sugar or methane into high‑value chemicals—bioplastics, pharmaceuticals, and even carbon‑neutral fuels—by leveraging carbon’s ability to form diverse molecular scaffolds. This bio‑manufacturing paradigm reduces reliance on petrochemical processes and minimizes waste Small thing, real impact..
In space exploration, carbon’s lightweight yet solid nature makes it a prime candidate for constructing habitats and tools on other planetary bodies. Consider this: researchers are investigating in‑situ resource utilization (ISRU) techniques that would extract carbon from Martian regolith or lunar soil to fabricate building blocks via 3D printing. Carbon‑based composites could provide radiation shielding and structural integrity for habitats, while carbon nanotube‑reinforced materials could enable stronger, lighter spacecraft components Small thing, real impact..
The Future Outlook: Harnessing Carbon Responsibly
The trajectory of carbon research points toward a future where the element’s full potential is realized responsibly. Advances in computational chemistry and machine learning are accelerating the discovery of novel carbon‑based materials, predicting properties before they are synthesized in the lab. Meanwhile, interdisciplinary collaborations among chemists, engineers, ecologists, and policymakers are essential to make sure carbon technologies align with sustainability goals Which is the point..
Key challenges remain: scaling up carbon capture technologies to gigatonne levels, ensuring the environmental safety of nanocarbon materials, and transitioning economies away from fossil‑derived carbon while maintaining energy security. Addressing these issues will require not only scientific ingenuity but also societal commitment to equitable and climate‑conscious solutions Nothing fancy..
Easier said than done, but still worth knowing Worth keeping that in mind..
Conclusion
Carbon’s story is one of paradox and promise—a simple atom whose six protons give rise to a complexity that underpins life, fuels economies, and drives innovation. From the microscopic bonds that stitch together DNA to the macroscopic structures of diamonds and graphene, carbon’s versatility is the thread that weaves together the natural world and human achievement. As we deepen our understanding of its behavior and develop technologies that respect the delicate balance of the carbon cycle, we stand poised to harness this elemental marvel for the betterment of both our planet and our future among the stars. The continued exploration of carbon chemistry is, therefore, not merely an academic pursuit; it is a vital endeavor that will shape the trajectory of life on Earth and beyond Small thing, real impact. Simple as that..