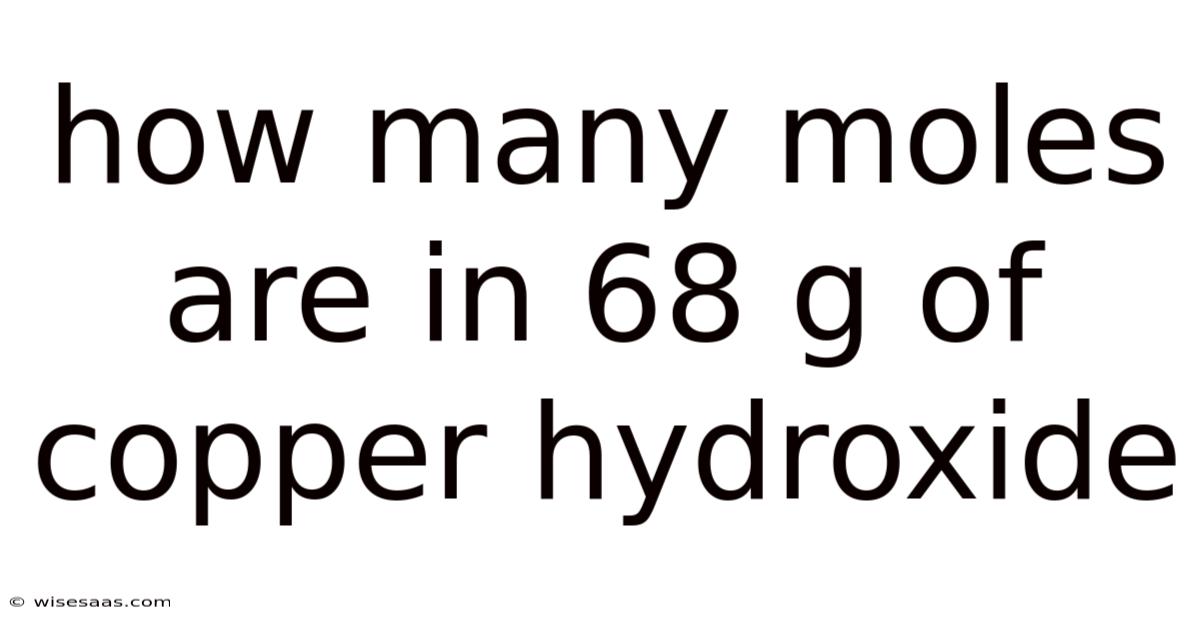How Many Moles Are in 68 g of Copper Hydroxide?
Copper hydroxide (Cu(OH)₂) is a chemical compound commonly used in industrial and laboratory settings. If you’re working with 68 grams of copper hydroxide and need to determine the number of moles present, the process involves understanding molar mass, stoichiometry, and unit conversions. This article will guide you through the calculation step by step, explain the science behind it, and address common questions to ensure clarity Worth keeping that in mind..
Understanding Moles and Molar Mass
Before diving into the calculation, it’s essential to grasp the foundational concepts:
- Mole: A mole is a unit of measurement in chemistry that represents 6.022 × 10²³ particles (Avogadro’s number). It allows chemists to count atoms or molecules in a practical way.
- Molar Mass: The molar mass of a compound is the mass (in grams) of one mole of that substance. It is calculated by summing the atomic masses of all atoms in the compound’s chemical formula.
For copper hydroxide (Cu(OH)₂), the molar mass depends on the atomic masses of copper (Cu), oxygen (O), and hydrogen (H) The details matter here. Surprisingly effective..
Step-by-Step Calculation
1. Determine the Chemical Formula
Copper hydroxide has the formula Cu(OH)₂. This means:
- 1 atom of copper (Cu)
- 2 atoms of oxygen (O)
- 2 atoms of hydrogen (H)
2. Find the Atomic Masses
Using the periodic table:
- Copper (Cu): 63.55 g/mol
- Oxygen (O): 16.00 g/mol
- Hydrogen (H): 1.008 g/mol
3. Calculate the Molar Mass of Cu(OH)₂
Multiply the atomic mass of each element by the number of atoms in the formula and sum the results:
$
\text{Molar Mass} = (1 \times 63.55) + (2 \times 16.00) + (2 \times 1.008)
$
$
= 63.55 + 32.00 + 2.016 = 97.566 , \text{g/mol}
$
4. Use the Formula to Convert Grams to Moles
The formula for converting mass to moles is:
$
\text{Moles} = \frac{\text{Mass (g)}}{\text{Molar Mass (g/mol)}}
$
Plugging in the values:
$
\text{Moles of Cu(OH)₂} = \frac{68 , \text{g}}{97.566 , \text{g/mol}} \approx 0.697 , \text{mol}
$
Scientific Explanation
This calculation relies on dimensional analysis, a method that ensures units cancel out to give the desired result. Here’s why it works:
- The molar mass acts as a conversion factor between grams and moles.
566 g units are in 68 g?Practically speaking, - By dividing the given mass (68 g) by the molar mass (97. 566 g/mol), you’re essentially asking, “How many 97.” The answer is the number of moles.
This principle is critical in stoichiometry, where precise measurements are necessary for chemical reactions, such as determining reactant ratios or predicting product yields Practical, not theoretical..
Common Mistakes to Avoid
- Incorrect Molar Mass: Forgetting to account for all atoms in the formula (e.g., miscounting the subscript “2” in Cu(OH)₂).
- Rounding Errors: Using rounded atomic masses
Conclusion
The calculation of moles from a given mass, as demonstrated with copper hydroxide (Cu(OH)₂), underscores the importance of precision in chemical measurements. This leads to by understanding the relationship between mass and moles through molar mass, chemists can accurately determine quantities of substances involved in reactions, ensuring reliable results in experiments and industrial applications. Day to day, this process not only reinforces fundamental concepts like Avogadro’s number and dimensional analysis but also highlights the practical utility of stoichiometry in solving real-world problems. That said, whether in a laboratory setting or theoretical studies, mastering these calculations is essential for advancing scientific knowledge and achieving accurate outcomes. The bottom line: the ability to convert between mass and moles empowers chemists to work efficiently, minimize errors, and apply chemical principles effectively across diverse fields.
Worth pausing on this one It's one of those things that adds up..
Practical Applications in the Laboratory and Industry
Once the number of moles has been established, the value becomes a gateway to a wide array of downstream calculations. In analytical chemistry, for instance, the mole count is used to prepare standard solutions with known concentrations, a prerequisite for titrations, spectroscopy, and chromatography. By weighing an exact mass of Cu(OH)₂ and converting it to moles, a technician can dissolve the sample in a defined volume of solvent, yielding a solution whose concentration is precisely known. This step is vital when calibrating instruments or verifying the purity of a commercial reagent.
In synthetic chemistry, stoichiometric relationships dictate the theoretical yield of a reaction. On the flip side, knowing that one mole of Cu(OH)₂ will react with, say, two moles of an acid to produce copper(II) salt and water allows the chemist to predict how much product can be formed from a given amount of reactant. If a laboratory experiment calls for the precipitation of copper(II) hydroxide from a copper sulfate solution, the calculated mole value (≈ 0.697 mol) informs the exact volume of acid needed to drive the reaction to completion, thereby optimizing reagent use and minimizing waste.
Industrial processes that involve bulk production of copper compounds rely on the same conversion principle, albeit on a much larger scale. Plant operators must translate kilogram‑scale feedstocks into mole quantities to control reaction conditions, maintain product specifications, and ensure safety. Here's one way to look at it: when producing copper(II) oxide by heating Cu(OH)₂, the mole figure determines the amount of thermal energy required and the expected mass of the final oxide, enabling efficient furnace loading and emissions monitoring Surprisingly effective..
Error Propagation and Significant Figures
Because the final answer is derived from measured mass (68 g) and a molar mass that itself is the sum of several atomic weights, any uncertainty in those inputs will affect the mole result. Propagating the uncertainty through the division step yields an approximate ±0.But 01 mol error for the example, assuming the mass measurement is accurate to ±0. 1 g. Reporting the answer with the appropriate number of significant figures—here, three—communicates the reliability of the calculation and aligns with best laboratory practice.
Linking Theory to Real‑World Outcomes
The ability to translate a macroscopic mass into a microscopic count of particles is more than an academic exercise; it bridges the gap between what can be weighed on a balance and what occurs at the atomic level during a chemical transformation. This bridge enables chemists to:
- Design reproducible experiments where the same amount of material is used each time, leading to consistent and comparable results.
- Scale up reactions from bench‑top to pilot‑plant or manufacturing levels, preserving the same stoichiometric relationships.
- Diagnose and troubleshoot unexpected outcomes by checking whether the calculated mole ratios align with observed product yields.
Final Synthesis
Boiling it down, converting a measured mass of a compound such as copper hydroxide into moles is a foundational skill that underpins quantitative chemistry. Now, by accurately determining molar mass, applying dimensional analysis, and interpreting the resulting mole value, scientists can predict reaction behavior, control experimental conditions, and translate laboratory discoveries into practical applications. Mastery of this conversion not only enhances precision in the lab but also empowers engineers and manufacturers to operate safely and efficiently at scale, ultimately advancing both scientific understanding and technological innovation.
Looking Ahead: Digital Integration and Sustainable Practices
The next wave of chemical engineering will embed real‑time analytics into every stage of production. Even so, spectroscopic sensors coupled with machine‑learning algorithms can automatically recalculate mole balances on the fly, adjusting feed rates before deviations translate into waste or safety hazards. Such closed‑loop control reduces the reliance on manual gravimetric checks, freeing technicians to focus on process optimization rather than repetitive calculations Small thing, real impact. Nothing fancy..
Simultaneously, the industry is gravitating toward greener chemistries that minimize the mass of reagents required for a given output. Also, by selecting pathways with higher atom‑economy, manufacturers can lower the total mass introduced into a reactor, which in turn lessens the energy burden associated with heating, cooling, and waste treatment. In this context, the mole concept becomes a strategic lever: a smaller, more efficient mole balance directly translates into reduced carbon footprints and cost savings Turns out it matters..
Education and Collaborative Innovation
Academic curricula are evolving to reflect these shifts. On the flip side, laboratory courses now integrate spreadsheet‑based simulations and Python notebooks that let students visualize how variations in molar mass or measurement error cascade through stoichiometric calculations. Collaborative platforms enable cross‑institutional projects where students from chemistry, materials science, and data engineering co‑author papers that explore, for instance, how nanoconfined copper oxides behave differently when the precursor is dosed in moles versus grams.
These interdisciplinary experiences cultivate a workforce fluent in both the language of atoms and the vocabulary of data analytics—an essential competency for the challenges that lie ahead Easy to understand, harder to ignore..
Conclusion
From the balance‑scale weighing of a few grams to the high‑throughput synthesis of copper‑based catalysts that power renewable‑energy technologies, the conversion of mass to moles remains the linchpin that connects observable reality with the invisible world of atoms. Mastery of this conversion equips chemists, engineers, and manufacturers with a precise, reliable tool for designing reactions, controlling scale‑up, and ensuring safety. As digital monitoring, sustainable chemistry, and interdisciplinary education reshape the landscape, the fundamental principle—dividing a measured mass by a compound’s molar mass—will continue to serve as the quantitative backbone of scientific progress, driving innovation while honoring the responsibility to protect both people and the planet.