Hand Sanitizing Gel Should Be At Least _______ Percent Alcohol.
wisesaas
Mar 13, 2026 · 4 min read
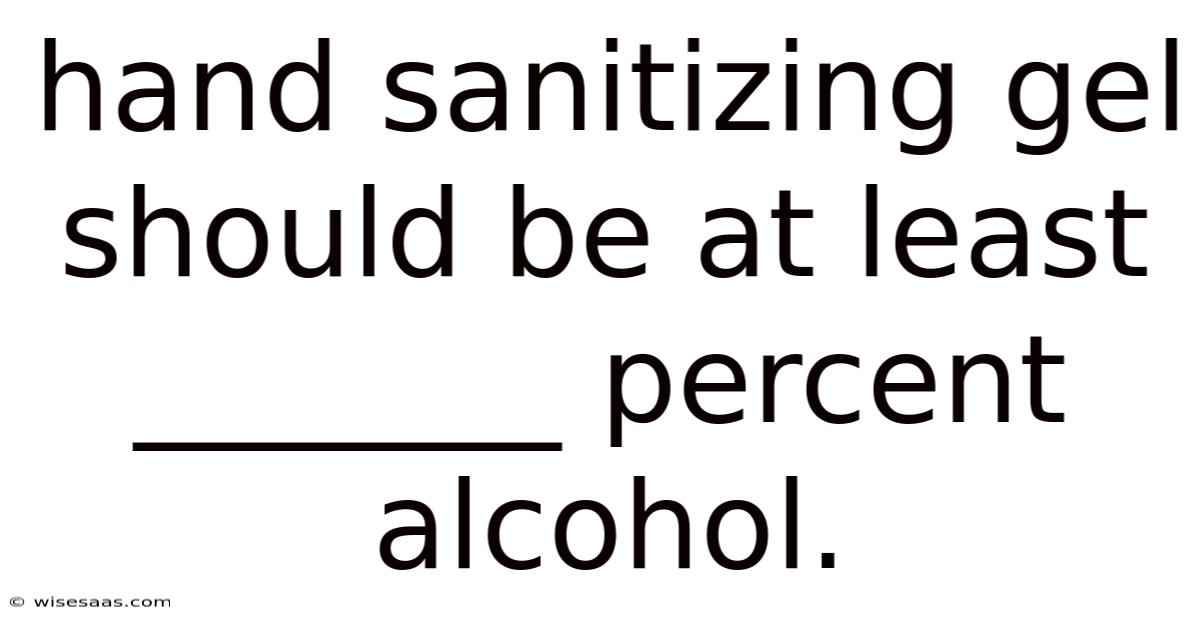
Table of Contents
Hand Sanitizing Gel Should Be At Least 60 Percent Alcohol
In the post-pandemic world, a small bottle of hand sanitizer has become as essential as keys or a wallet. Yet, many consumers remain unaware of the single most critical factor determining its effectiveness: the alcohol concentration. Hand sanitizing gel should be at least 60 percent alcohol to reliably kill the vast majority of harmful germs, bacteria, and viruses that pose a risk to human health. This isn't a arbitrary marketing figure; it is a scientifically established minimum threshold endorsed by global health authorities like the Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO). Choosing a product with less than 60% alcohol is not just a lesser choice—it can render the sanitizer virtually useless, providing a dangerous false sense of security. Understanding this percentage is key to making an informed decision that protects you and your family.
The Critical Threshold: Why 60% is the Magic Number
The 60% alcohol benchmark is the point at which a sanitizer’s germ-killing power becomes both potent and reliable. This concentration applies to the two primary active ingredients: ethanol (ethyl alcohol) and isopropyl alcohol (rubbing alcohol). Products falling within the 60% to 95% alcohol range are considered clinically effective for hand hygiene when soap and water are not available.
- Below 60%: Sanitizers with an alcohol content below 60% lack the necessary punch to denature the proteins of many microbes effectively. They may reduce the number of germs but will not eliminate them consistently, especially more resilient pathogens. This "sub-therapeutic" level can allow some viruses and bacteria to survive and even potentially develop resistance over time.
- The Optimal Range (60%-95%): Within this band, the alcohol content is high enough to break down the protective outer layers of microbes and coagulate their internal proteins, leading to rapid cell death. Water is a crucial component in this mixture; it facilitates the denaturation process and prevents the alcohol from evaporating too quickly, ensuring sufficient contact time with the germs.
- Above 95%: Interestingly, concentrations exceeding 95% (like pure ethanol) are actually less effective. Without water, the alcohol coagulates the outer protein layer of a microbe too quickly, creating a hardened shell that protects the inner cell, preventing the alcohol from penetrating and destroying it completely. This is why the most effective formulations balance alcohol with water and other humectants.
The Science of Sanitization: How Alcohol Kills Germs
To appreciate why the percentage is so vital, it helps to understand the mechanism. Alcohol is a disinfectant that works through a process called denaturation. When applied to the hands, the alcohol molecules:
- Disrupt Lipid Membranes: Many bacteria and all enveloped viruses (like influenza, SARS-CoV-2, and HIV) have a fatty lipid outer membrane. Alcohol is a solvent that dissolves this protective layer, causing the microbe’s insides to leak out.
- Denature Proteins: Alcohol molecules infiltrate the microbe’s structure and break the hydrogen bonds that hold its essential proteins in their functional shape. Once these proteins are misshapen (denatured), the microbe cannot perform vital life functions and dies.
- Require Contact Time: For this process to be complete, the sanitizer must remain wet on the skin for about 20-30 seconds. A gel or rub with the correct alcohol concentration stays active long enough to do its job before evaporating.
This mechanism is highly effective against a broad spectrum of pathogens, including:
- Enveloped Viruses: COVID-19, common cold viruses, herpes, measles.
- Many Bacteria: E. coli, Salmonella, Staphylococcus aureus (including some MRSA strains).
- Some Fungi: Like the yeast that causes athlete’s foot.
However, it is important to note that alcohol-based sanitizers do not kill all germs. They are ineffective against non-enveloped viruses (such as norovirus and rotavirus), bacterial spores (like C. difficile), and certain protozoan parasites. For these, thorough handwashing with soap and water remains the gold standard, as soap physically lifts and rinses away these resilient organisms.
Dangers of Low-Alcohol and "Natural" Alternatives
The market is flooded with products that cleverly skirt the 60% rule. These often appeal to consumers seeking "gentler," "natural," or "child-friendly" options but can be dangerously ineffective.
- Alcohol-Free Sanitizers: These typically use benzalkonium chloride as the active ingredient. While it is a registered disinfectant, studies have shown it to be significantly less effective than alcohol against many viruses, including coronaviruses. Its efficacy is also more variable and can be neutralized by organic material (dirt, food residue) more easily.
Latest Posts
Latest Posts
-
What Type Of Writing Is Most Similar To An Autobiography
Mar 13, 2026
-
What Are The Three Components Of An Effective Argument
Mar 13, 2026
-
Which Of The Following Is A Non Renewable Energy Source
Mar 13, 2026
-
The Honeycomb Like Appearance Of This Sandstone Is A Result Of
Mar 13, 2026
-
Which Statement Would A Humanist Most Likely Agree With
Mar 13, 2026
Related Post
Thank you for visiting our website which covers about Hand Sanitizing Gel Should Be At Least _______ Percent Alcohol. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.