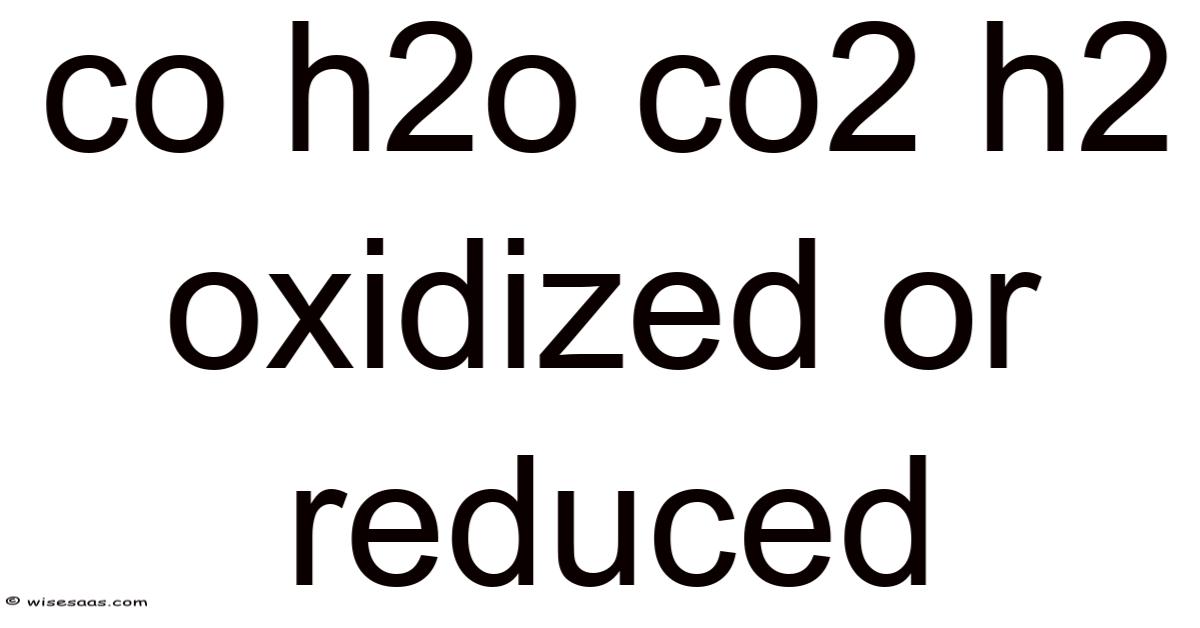Understanding Oxidation and Reduction in CO, H2O, and CO2
Oxidation and reduction are fundamental concepts in chemistry that describe the transfer of electrons between substances. To determine whether carbon in CO, H2O, and CO2 is oxidized or reduced, we must analyze the oxidation states of the elements involved. Oxidation states, also known as oxidation numbers, indicate the hypothetical charge an atom would have if all bonds were ionic. These processes are central to redox reactions, which are essential in biological systems, industrial processes, and environmental chemistry. By comparing these values, we can identify whether a substance has lost or gained electrons Worth keeping that in mind..
What is Oxidation and Reduction?
Oxidation refers to the loss of electrons by an atom, while reduction refers to the gain of electrons. In a redox reaction, one substance is oxidized, and another is reduced. The oxidation state of an atom helps determine this:
- A positive oxidation state indicates the atom has lost electrons (oxidized).
- A negative oxidation state indicates the atom has gained electrons (reduced).
To give you an idea, in the reaction between hydrogen and oxygen to form water (H2 + O2 → H2O), hydrogen is oxidized (from 0 to +1), and oxygen is reduced (from 0 to -2) And that's really what it comes down to..
Oxidation States in CO, H2O, and CO2
1. Carbon Monoxide (CO)
Carbon monoxide (CO) is a molecule composed of one carbon atom and one oxygen atom. To determine the oxidation state of carbon:
- Oxygen typically has an oxidation state of -2 in most compounds.
- The compound is neutral, so the sum of oxidation states must equal 0.
Let the oxidation state of carbon be x.
$
x + (-2) = 0 \implies x = +2
$
Thus, the oxidation state of carbon in CO is +2. Since carbon in its elemental form (C) has an oxidation state of 0, the carbon in CO has lost electrons (from 0 to +2). This means carbon in CO is oxidized.
2. Carbon Dioxide (CO2)
Carbon dioxide (CO2) contains one carbon atom and two oxygen atoms. Again, oxygen has an oxidation state of -2. The compound is neutral, so:
Let the oxidation state of carbon be x.
$
x + 2(-2) = 0 \implies x = +4
$
The oxidation state of carbon in CO2 is +4. Compared to its state in CO (+2), carbon has lost more electrons (from +2 to +4). This indicates that carbon in CO2 is further oxidized than in CO It's one of those things that adds up..
3. Water (H2O)
Water (H2O) is composed of two hydrogen atoms and one oxygen atom. That said, carbon is not present in H2O, so it is not relevant to the oxidation or reduction of carbon. This might be a point of confusion, as the question includes H2O. If the intended compound was hydrogen peroxide (H2O2), the analysis would differ. To give you an idea, in H2O2, oxygen has an oxidation state of -1, and hydrogen is +1. But since the question specifies H2O, we focus on the compounds containing carbon And that's really what it comes down to..
Comparing Oxidation States Across Compounds
To determine whether carbon is oxidized or reduced, we compare its oxidation states in different compounds:
- CO: Carbon is +2 (oxidized compared to elemental carbon, which is 0).
- CO2: Carbon is +4 (more oxidized than in CO).
- H2O: No carbon, so no oxidation or reduction of carbon occurs.
This shows that carbon in CO and CO2 is oxidized, with CO2 being more oxidized than CO.
Why Does Oxidation State Matter?
Oxidation states are critical for understanding chemical reactions. Here's a good example: in combustion reactions, carbon in hydrocarbons (like methane, CH4) is oxidized to CO2. The oxidation state of carbon increases from -4 in CH4 to +4 in CO2, indicating a significant loss of electrons. Similarly, in photosynthesis, carbon dioxide (CO2) is reduced to form glucose (C6H12O6), where carbon’s oxidation state decreases from +4 to 0 Less friction, more output..
Common Misconceptions
- H2O and Carbon: Since H2O contains no carbon, it cannot be involved in the oxidation or reduction of carbon. This might be a typo or misunderstanding in the question.
- **